|
No, the chlorate ion does not have a 2 charge. This results in a net negative charge of -1 on the molecule, making it an anion (a negatively charged ion). This means that the electron distribution within the molecule is asymmetrical, with more electrons being attracted to the chlorine atom than the oxygen atoms. The ClO3 molecule has a negative charge because the chlorine atom has a higher electronegativity than the oxygen atoms. This makes it useful for many industrial applications such as bleaches, detergents, matches, gunpowder, rocket fuel and water treatment processes. In conclusion, the chlorate ion carries a negative electrical charge due to its difference in electronegativity between chlorine and oxygen atoms. 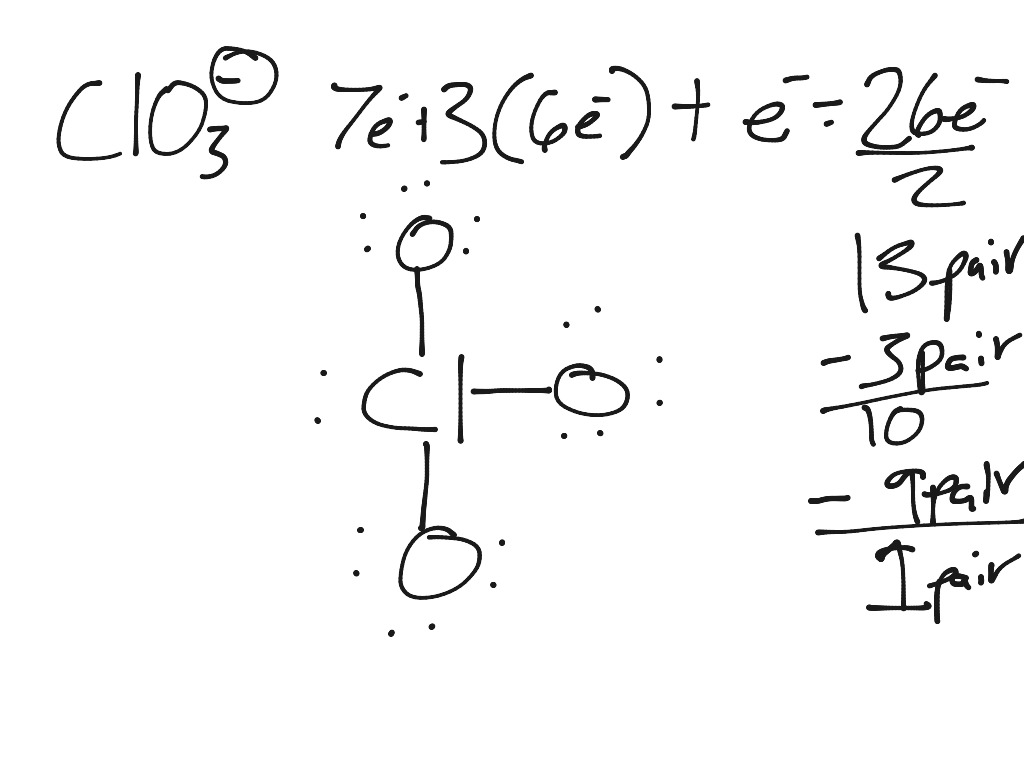
It can also be used to produce chlorine dioxide which is a powerful bleaching agent used in paper mills and textile mills. 
It is widely used in industries such as paper manufacturing, metalworking and water treatment as it has strong oxidizing properties which make it useful for removing impurities from wastewater. The most common chlorate is sodium chlorate which is produced by electrolysis of sodium chloride (table salt) solution. Chlorates can also be used in agriculture as a fertilizer or herbicide. These compounds are referred to as chlorates and are often used in industrial applications such as bleaches, detergents, matches, gunpowder and rocket fuel. The shared electrons are not located in a fixed position between the nuclei.Due to its negative charge, chlorate can form compounds with oher elements and ions. In Lewis structures the bonding pair of electrons is usually displayed as a line, and the unshared electrons as dots: When two chlorine atoms covalently bond to form \(Cl_2Cl_2\), the following sharing of electrons occurs:Įach chlorine atom shared the bonding pair of electrons and achieves the electron configuration of the noble gas argon. In a sense, it has the electron configuration of the noble gas helium. The shared pair of electrons provides each hydrogen atom with two electrons in its valence shell (the 1 s) orbital. The diatomic hydrogen molecule (H 2) is the simplest model of a covalent bond, and is represented in Lewis structures as: Although it is said that atoms share electrons when they form covalent bonds, they do not usually share the electrons equally. Nonmetals will readily form covalent bonds with other nonmetals in order to obtain stability, and can form anywhere between one to three covalent bonds with other nonmetals depending on how many valence electrons they posses. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. The arrow indicates the transfer of the electron from sodium to chlorine to form the Na metal ion and the Cl - chloride ionĬovalent bonding occurs when pairs of electrons are shared by atoms. 
The Lewis Structure of this reaction is (here we will consider one chlorine atom, rather than Cl 2) is: 
Thus the chlorine gains an electron from the sodium atom. The chlorine has a high affinity for electrons, and the sodium has a low ionization potential.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
